Nitra Force
NBR Nitrile Examination gloves
Nitra Force
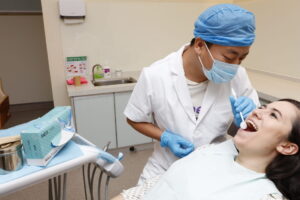
The perfect Nitrile Examination gloves! Gloves are made of nitrile for their good wearing and comfort and are often used as an alternative to latex gloves as they do not contain any latex proteins which causes allergies. The gloves are extremely comfortable and have high tear resistance due to their elasticity and flexibility.
Nitra Force Nitrile examination gloves can be put on and taken off easily without powder due to their smooth inner surface. Not only do the gloves have exceptional wearing properties but Nitra Force gloves will provide you with the highest protection. The gloves are resistant to bacteria, viruses, and chemicals; and are even permeable to some solvents such as acetone! Providing full protection with a perfect fit.



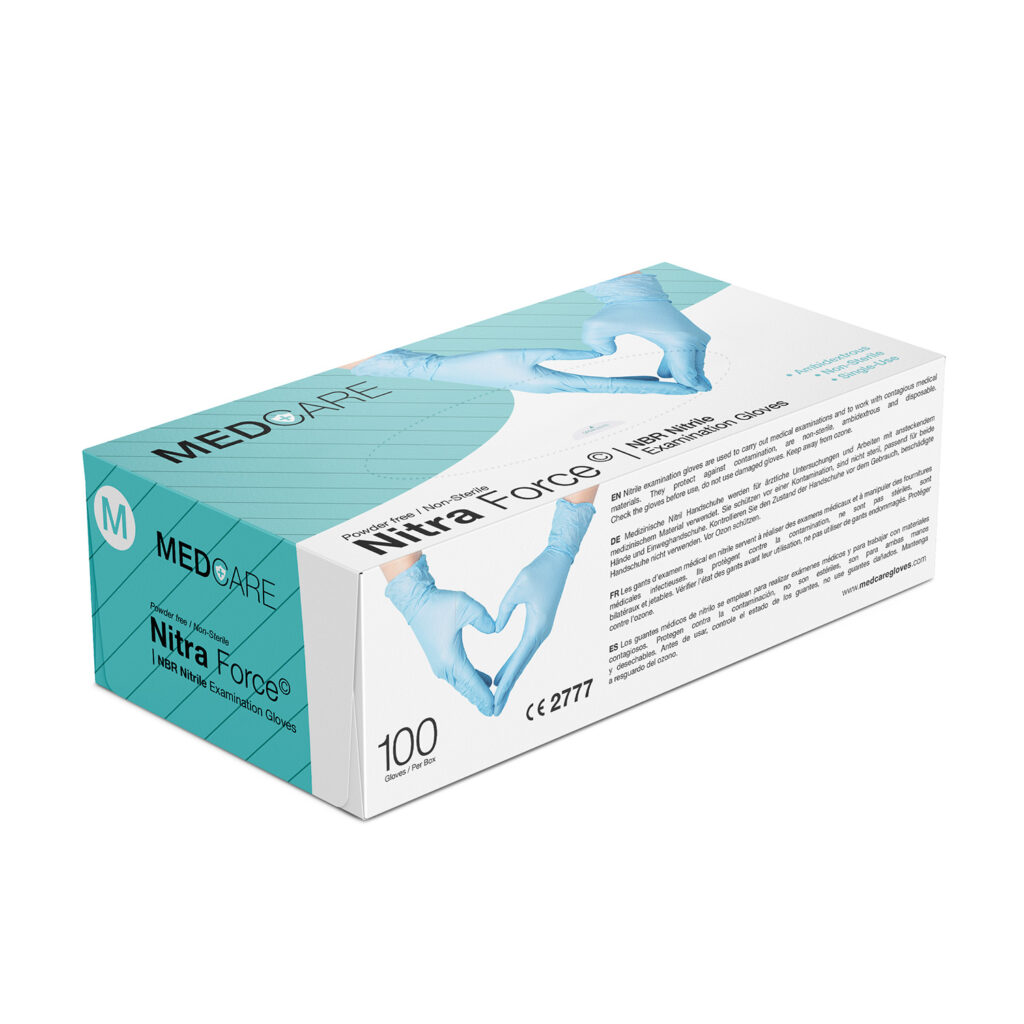
- Choose color variant





Features
- Inspected to AQL 1.5 – Acceptable Quality Level
- Non-sterile
- Free from natural latex and latex proteins
- Powder-free
- Ambidextrous
- Single-use
- Blue, Light Blue, Pink, Purple, Black colors
- Very good skin compatibility
- Relatively high wall thickness for greater resistance to chemical effects
- High impermeability to viruses and bacteria
- Available sizes S, M, L, XL
- Complies with MDR (EU) 2017/745 (Class I) and PPE (EU) 2016/425 (Category III)
- Suitable for food processing
Harmonised standards
EN ISO 13485:2016, EN ISO 9001:2015, EN ISO 14971:2019, IEC 62366-1:2015(en), EN ISO 15223-1:2016, EN 455-1:2000, EN 455-2:2015, EN 455-3:2015, EN 16523-1:2015+A1:2018, EN ISO 374-1:2016+A1:2018, EN 420:2003+A1:2009, EN 374-2:2014, EN ISO 374-4:2019, EN ISO 374-5:2016, EN 1186-1:2002, EN 1186-3:2002, EN 1186-14:2002, BS EN 13130-1:2004, BS EN 12868:1999
ASTM D6124:06(2017)
Standard Test Method for Residual Powder on Medical Gloves
ASTM 6319
Standard Specification for Nitrile Examination Gloves for Medical Application
ASTM D5151-19
Standard Test Method for Detection of Holes in Medical Gloves
ASTM D6978-05(2019)
Standard Practice for Assessment of Resistance of Medical Gloves to Permeation by Chemotherapy Drugs
Personal protective equipment certification. Notified Body responsible for certification and ongoing conformity (SATRA Technology Europe Ltd.)
Mecare Nitra Force gloves are manufactured and labeled in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council on medical devices and Regulation (EU) 2016/425 of the European Parliament and of the Council on personal protective equipment and that this medical device and personal protective equipment is for the intended purpose of use safe, effective, and suitable for the provision of health care. The personal protective equipment type-examination (module B) was performed by the notified body SATRA Technology Europe Ltd. (2777). EU Type-Examination Certificate number: 2777/15456-02/E00-00.